#Ifactor cerapedics trial#
In March, Cerapedics said it won FDA investigational device exemption approval to launch a clinical trial examining the safety and efficacy of its P-15L Peptide Enhanced bone graft.įiled Under: Business/Financial News, Orthopedics Tagged With: Cerapedics Inc.

This financing will allow Cerapedics to further invest to increase physician access to i-Factor and to build clinical evidence for new indications,” Dr. “Cerapedics is well-positioned for strong growth with Level I data establishing i-Factor’s superiority and with a dedicated team that has deep experience in orthobiologics and spinal fusion. In addition to the round, Cerapedics said it added KCK group medical tech division managing director Valeska Schroeder, KCK medical tech division head Greg Garfield and KCK investment partner Nael Kassar. Manuel with a solid background in banking and Cosmin as an entrepreneur, are familiar with the difficulties that SMEs face when looking to access the necessary working capital from banks. We are fortunate to be supported by KCK Group, a partner that shares in the long-term vision that differentiated products that provide safety, efficacy and economic value, supported by human clinical results, will ultimately provide the highest value to surgeons and their patients,” CEO Glen Kashuba said in a press release. At the heart of our initiative is innovation, transforming iFactor into a solution out of the traditional patterns of financing methods. We also understand the value of Level I human clinical data and are committed to investing and expanding our indications. This year we have exceeded our own expectations regarding year over year revenue growth and have significantly expanded our user base as surgeons continue to express a high level of satisfaction with clinical results that support the safety and efficacy of our product. In November 2015, Cerapedics received Premarket Approval (PMA) from the FDA for the use of i-FACTOR Bone Graft in ACDF procedures.
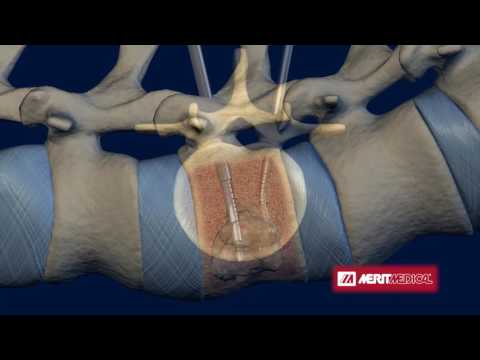
“We are pleased to announce that we have successfully completed an equity financing that will continue to accelerate the commercial release of i-Factor Peptide enhanced bone graft and support the execution of a second Level I investigational device exemption human clinical study that will broaden our market indication in the $2 billion U.S. iFACTOR Bone Graft is injected during spinal fusion surgeries. iFACTOR Bone Graft is based on synthetic small peptide (P-15) technology developed by Cerapedics to support bone growth through cell attachment and activation.
The funding round was led by family investment fund KCK Group, the Westminster, Colo.-based company said, and will help support the company’s bone graft products. Contact a supplier or the parent company directly to get. Cerapedics said late last week it closed a $22 million round of financing and added three new members to its board of directors Cerapedics initial P-15 technology product, i-FACTOR Peptide Enhanced Bone Graft, was approved in the European Union in 2008 and Australia in 2010 for use in the repair of bony voids or defects in orthopedic applications throughout the skeletal system (i.e., the spine and extremities). Find out all of the information about the Cerapedics product: synthetic bone substitute i-FACTOR.
